
To contact the author of this article, email Siobhan.Treacy@ieeeglobalspec.com
In energy production, energy transition is dependent on technologies that boast cheap and temporary storage of renewable energy. Researchers have found that aluminum batteries are one of the most promising candidates to fill this need because they are made from cheap and abundant raw materials and can hold a good amount of renewable energy.
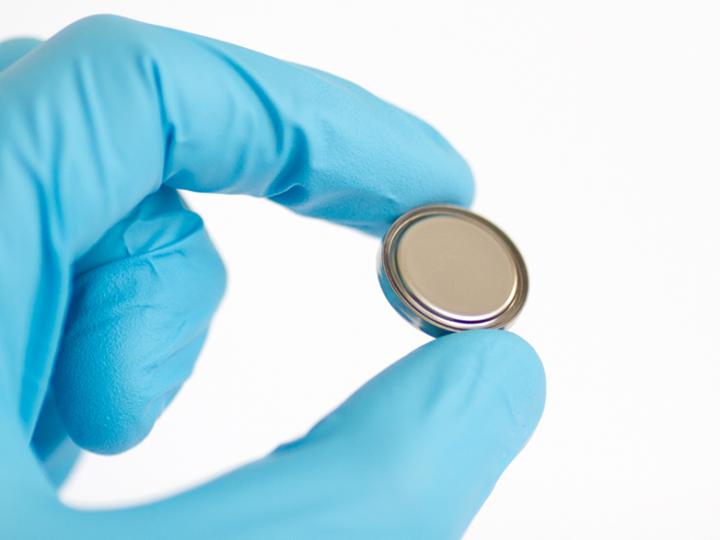 The researchers produced aluminum button cells in the laboratory. The battery case is made of stainless steel coated with titanium nitride on the inside to make it corrosion resistant. Source: ETH Zurich / Kostiantyn Kravchyk
The researchers produced aluminum button cells in the laboratory. The battery case is made of stainless steel coated with titanium nitride on the inside to make it corrosion resistant. Source: ETH Zurich / Kostiantyn Kravchyk
The scientists and researchers from ETH Zurich and Empa were led by Makysm Kovalenko, Professor of Functional Inorganic Materials. This team is just a small number of the researchers worldwide who are focusing on developing these kinds of batteries. The team found two new materials that could be huge for the development of aluminum batteries. The first, a corrosion-resistant material, would be used in the conductive parts of the battery. The second is a novel material that would be used on the positive pole and that can be adapted for many technical requirements.
The electrolyte fluid in aluminum batteries is aggressive and corrodes many materials like steel, gold and platinum. Researchers have been seeking corrosion-resistant materials that would work for the conductive areas of the batteries. Kovalenko’s team found the answers in titanium nitride, a ceramic material that exhibits high conductivity.
“This compound is made up of the highly abundant elements titanium and nitrogen, and it’s easy to manufacture,” explains Kovalenko.
Aluminum batteries with conductive parts made out of titanium nitride were successfully made in the team’s lab. The material is easily produced in a thin film or as a coating over other materials like polymer foils. Kovalenko says it could be possible to manufacture conductors from a conventional metal coated with titanium nitride or print conductive titanium nitride track on plastic.
“The potential applications of titanium nitride are not limited to aluminum batteries. The material could also be used in other types of batteries; for example, in those based on magnesium or sodium, or in high-voltage lithium-ion batteries,” says Kovalenko.
The second new material is polypyrene, a hydrocarbon with chain-like (polymeric) structure. Polypyrene is used for the positive electrode (pole) of aluminum batteries. The positive electrode in aluminum batteries is typically made out of graphite. The new material discovered by the team is competitive to graphite because it can hold more energy than graphite. The initial experiments show that the material is successful.
“A lot of space remains between the molecular chains. This allows the relatively large ions of the electrolyte fluid to penetrate and charge the electrode material easily,” Kovalenko explains.
Polypyrene allows scientists to influence its properties, adapting the materials for any specific application.
“In contrast, the graphite used at present is a mineral. From a chemical engineering perspective, it cannot be modified,” says Kovalenko.
Titanium nitride and polypyrene are flexible. The researchers say that the flexibility of the materials would be useful for “pouch cells”, which are batteries enclosed in a flexible film.
The new batteries would be used to store energy harvested from an eco-friendly source, like solar or wind, when these resources are unavailable. The batteries store this energy in a cost-effective way while being inexpensive and made out of abundant sources.
The paper on this research was published in Advanced Materials.

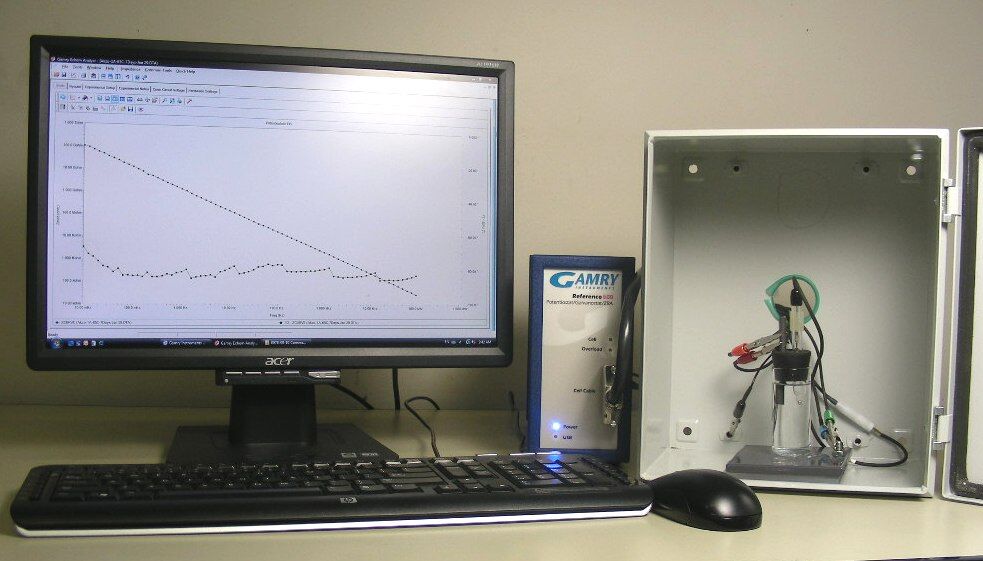 EIS testing apparatus. Photo courtesy of Amal Al-Borno, Charter Coating Service (2000), Ltd.
EIS testing apparatus. Photo courtesy of Amal Al-Borno, Charter Coating Service (2000), Ltd.


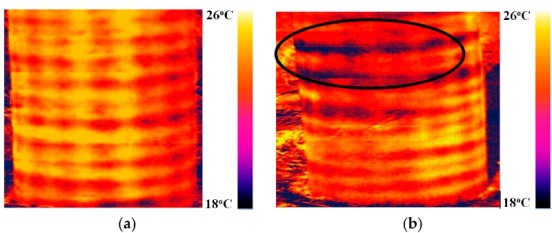
 The study was completed at Russia’s Trans-Siberian Railway, using an experimental facility for conducting induction and infrared thermal imaging. Photo courtesy of TPU.
The study was completed at Russia’s Trans-Siberian Railway, using an experimental facility for conducting induction and infrared thermal imaging. Photo courtesy of TPU.
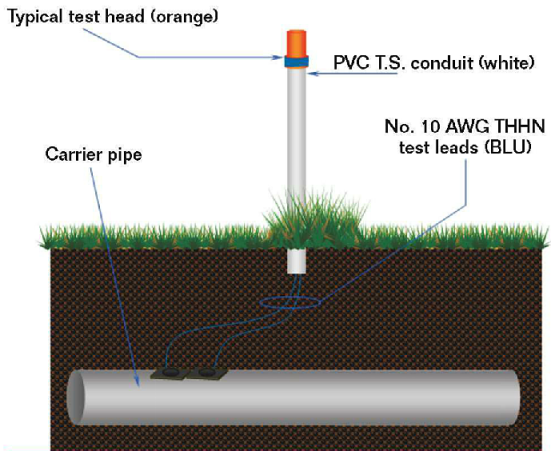
 During the installation of a new pipeline, corrosion engineers must consider many different aspects of the pipeline and its corrosion protection.
During the installation of a new pipeline, corrosion engineers must consider many different aspects of the pipeline and its corrosion protection.




 Tests were conducted on the chemical and mechanical properties of the graphene-added epoxy coatings, including the tensile strength. Photo courtesy of Talga Resources.
Tests were conducted on the chemical and mechanical properties of the graphene-added epoxy coatings, including the tensile strength. Photo courtesy of Talga Resources.




 The Weston Mil-Ammeter Model 264 was first patented in 1888.
The Weston Mil-Ammeter Model 264 was first patented in 1888.




